 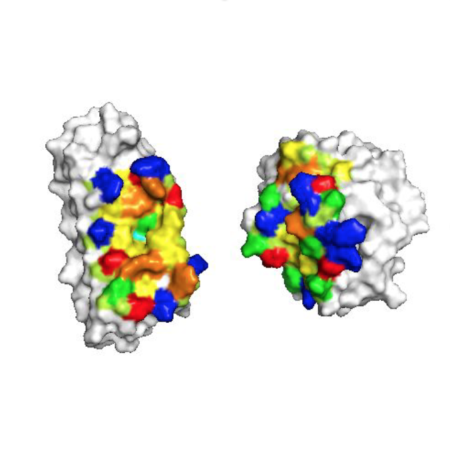
However, BH3-mimetics that non-specifically target multiple BCL2 proteins can cause harmful side effects by unnecessarily suppressing normal biological functions. Small molecule and peptide therapeutics mimic BOPs by binding pro-survival proteins, inducing apoptosis by disrupting inhibition of Bak and Bax and limiting sequestration of BOPs. Overexpression of one or more pro-survival homologs enables cancers to resist apoptosis, and different cancers have different profiles of pro-survival protein overexpression ( Kelly and Strasser, 2011 Placzek et al., 2010). Pathology arises when apoptosis is dysregulated. 
Designed inhibitors have a net pro-apoptotic effect by binding pro-survival proteins, which may both limit sequestration of BOPs ( A) and disrupt inhibition of Bak and Bax ( B). MOM permeabilization allows the release of cytochrome c and other factors from the intermembrane space (IMS) and thus initiates the apoptotic signaling cascade ( 7). Pro-survival proteins, which are typically overexpressed in cancer, bind and inhibit Bak and Bax ( 4), which would otherwise homo-oligomerize upon activation ( 5) and form pores in the mitochondrial outer membrane (MOM 6). BH3-only proteins (BOPs) are sequestered by pro-survival homologs (labels 1 and 2), and some BOPs may activate the direct effectors Bak and Bax by disrupting their inhibition by pro-survival proteins ( 3a) and/or promoting their homo-oligomerization ( 3b). The balanced network of interactions between pro-apoptotic and pro-survival members can be tipped toward cell death by cellular stress signals that induce transcription ( Essafi et al., 2005 Nakano and Vousden, 2001) or post-translational modification of BOPs ( Desagher et al., 2001 Fricker et al., 2010 reviewed in Shamas-Din et al., 2011).īCL2 proteins are categorized by their net effect on cell fate and the presence of shared structural domains. Interactions between BCL2 members are mediated by an amphipathic, helical BH3 motif that recognizes a conserved hydrophobic cleft present in the effectors and pro-survival proteins. BOPs can also activate effectors directly through transient binding interactions ( Dai et al., 2011 Kim et al., 2009 Walensky et al., 2006) or indirectly by binding pro-survival proteins and out-competing bound effectors ( Ku et al., 2011 Willis et al., 2007) or other direct activator BOPs ( Kuwana et al., 2005 Letai et al., 2002 Figure 1). Pro-survival homologs (six in humans: Bcl-2, Bcl-xL, Bcl-w, Mcl-1, Bfl-1 and Bcl-B) are structurally similar, but oppose apoptosis by binding and inhibiting Bak and Bax, as well as sequestering pro-apoptotic BH3-only proteins (BOPs). Pro-apoptotic effector proteins Bak and Bax have four distinct BH motifs and homo-oligomerize upon activation to form pores in the mitochondrial outer membrane, committing the cell to apoptosis. The key regulators of mitochondrial outer membrane permeability are B cell lymphoma-2 (BCL2) family proteins which are categorized functionally by their effect on cell fate, and structurally by the presence of BCL2 homology (BH) motifs. As cells enter apoptosis, mitochondrial membrane permeability increases, releasing mitochondrial factors such as cytochrome c that initiate destructive protease cascades in the cytosol. Programmed cell death is a tightly controlled process, involving both pro-survival and pro-apoptotic proteins that regulate permeability of the outer mitochondrial membrane. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
